Exploring the Innovations of Magstim in Neurotechnology.
Magstim has been doing cool stuff in the world of brain technology for a long time. They make devices that can interact with the brain and nerves without surgery. Think of it like a high-tech way to help people with different conditions, from pain to brain disorders. They’ve got a solid reputation and their equipment is used by researchers and doctors all over the place. It’s pretty interesting how these machines work and what they can do.
Key Takeaways
- Magstim has a long history of creating magnetic stimulation tools, trusted by many researchers and doctors globally.
- The Magstim Rapid system has new features for better performance and easier use, with flexible settings for different needs.
- Magstim Rapid has FDA clearance for treating chronic pain, offering a non-drug, non-invasive option.
- Magstim’s technology supports research into various brain conditions like depression, Alzheimer’s, and Parkinson’s.
- Magstim offers a range of neurotechnology solutions, including EEG systems, and works with other tools for a complete approach.
Magstim’s Pioneering Role in Neuromodulation
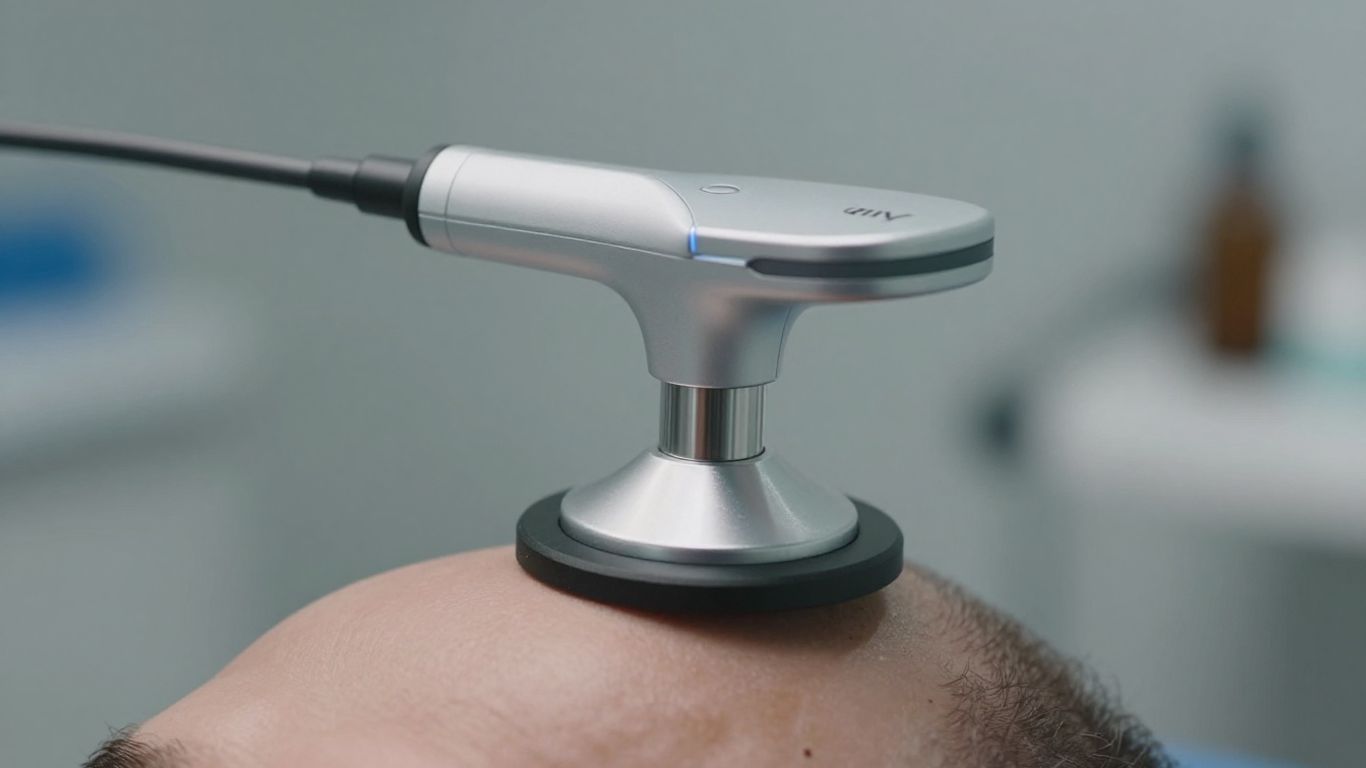
A Legacy of Innovation in Magnetic Stimulation
Magstim has been in the magnetic stimulation game for a long time, over 35 years to be exact. They’ve really been a big part of how we understand and use magnetic stimulation for both research and helping people. Think of them as one of the companies that helped get this whole field off the ground. Their equipment is used all over the world, in tons of studies – we’re talking over 15,000 scientific papers mention their tech. That kind of widespread use means a lot of doctors and scientists really trust what they make.
Trusted by Global Research and Clinical Communities
It’s not just a few people using Magstim; it’s a global community. Researchers in universities, doctors in hospitals, and clinicians in specialized clinics all rely on Magstim’s tools. This trust comes from years of reliable performance and technology that keeps up with scientific needs. When you see a company’s name pop up in so many research studies, it tells you they’re doing something right and that their equipment is dependable for serious work.
Shaping the Future of Non-Invasive Therapies
Magstim isn’t just about the present; they’re actively working on what’s next in non-invasive treatments. They’ve been instrumental in developing ways to interact with the nervous system without surgery or drugs. This focus on non-invasive methods is changing how we approach a lot of health issues, offering new hope for patients who might not have had many options before. Their work is paving the way for therapies that are safer and easier for patients to undergo.
The development of non-invasive neuromodulation techniques represents a significant shift in healthcare. By using magnetic fields to influence nerve activity, Magstim is contributing to a future where treatments can be more targeted, less burdensome, and accessible to a wider range of individuals seeking relief from various neurological and pain conditions.
Advancements in Magstim Rapid Technology
Next-Generation Features for Enhanced Performance
The Magstim Rapid system has seen some serious upgrades, making it even better for both research and clinical work. They’ve packed in new tech that really pushes what magnetic stimulation can do. The whole system is built to be more adaptable and perform better than ever before. It’s got a bigger, 15-inch touchscreen that makes it way easier to see what you’re doing, and the software is much more user-friendly with clearer steps to follow. Plus, managing patient data is now a smoother process, which is a big deal when you’re dealing with a lot of information.
Intuitive Software and Streamlined Data Management
One of the biggest changes is how easy the new software is to use. It’s designed with clear workflows, so you spend less time figuring out the controls and more time on the actual stimulation. This makes it simpler to set up sessions and track progress. The data management tools are also improved, helping you keep all your patient information organized and accessible. This is super important for research studies where accurate record-keeping is key. You can find out more about their advanced air-cooled TMS coil technology which helps keep treatments running smoothly.
Versatile Pulse Configurations for Diverse Applications
Magstim didn’t just update the interface; they also expanded the system’s capabilities. The Rapid now offers a wider range of pulse configurations to suit different needs. This includes:
- Standard repetitive TMS protocols
- Burst stimulation
- Biphasic pulse stimulation
These options come in different frequencies, like the Standard Rapid at 50Hz and the Super Rapid versions at 100Hz. This flexibility means the system can be used for a lot of different research areas and therapeutic applications, from studying brain pathways to treating various conditions. It’s this kind of adaptability that makes the Magstim Rapid a go-to tool in many labs and clinics.
FDA Clearance and Clinical Applications of Magstim
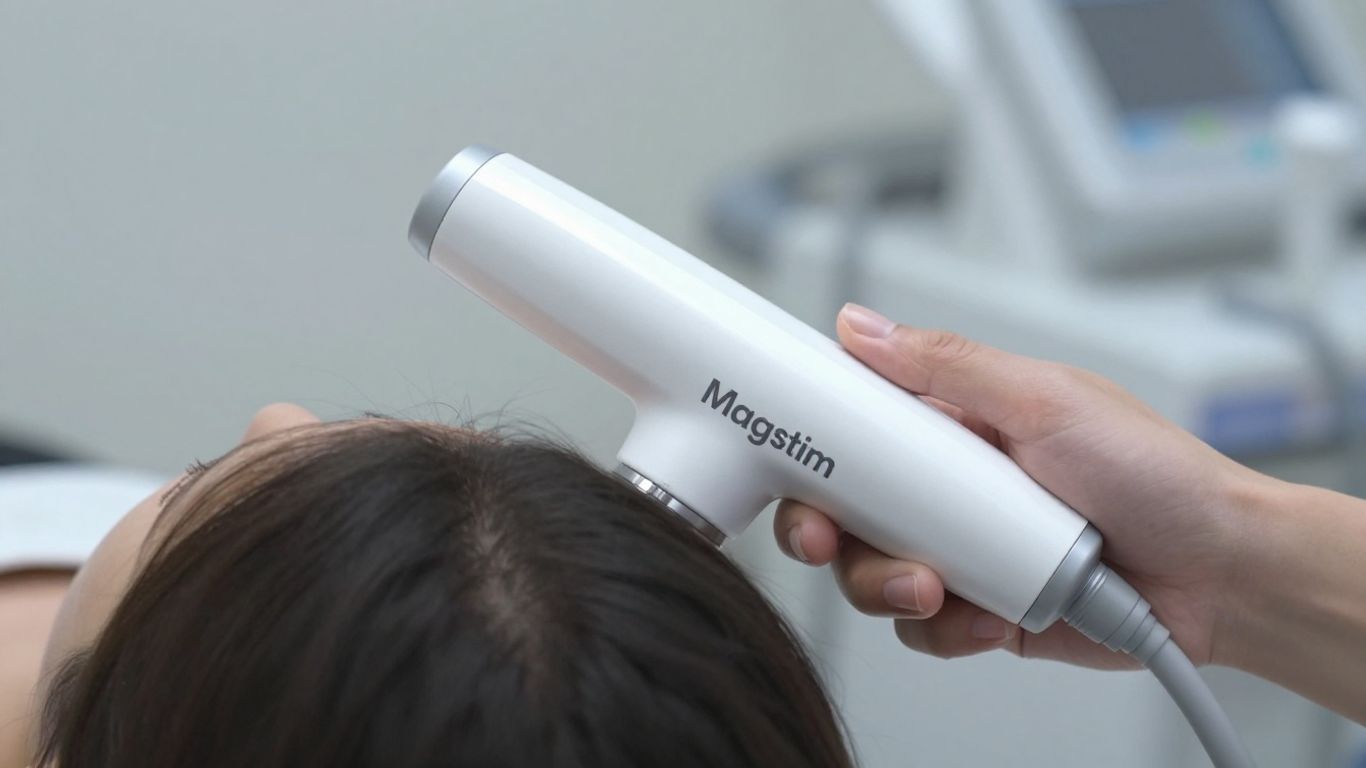
Magstim Rapid Cleared for Pain Management
It’s pretty big news that the Magstim Rapid system has received FDA clearance for treating chronic pain. This means a lot more people can now access a treatment that doesn’t involve drugs or surgery. It works by using magnetic pulses to interact with the nerves involved in how we feel pain. This clearance opens the door for a non-invasive, drug-free option for millions struggling with persistent discomfort.
Targeting Neural Pathways for Chronic Pain Relief
The Magstim Rapid system is designed to specifically target the nerve pathways that play a role in pain. It does this by placing a magnetic coil over a specific area, sending pulses that can reach nerves through the skin and soft tissues. The goal is to reduce pain intensity, help people move better, and generally improve their day-to-day lives. It’s a way to address pain at its source without needing implants or medications.
Expanding Access to Non-Drug, Non-Invasive Therapies
This development is a significant step forward in how we manage chronic pain. For a long time, options were limited, often involving medications with side effects or invasive procedures. Now, with FDA clearance, Magstim Rapid offers a validated, safe alternative. It’s particularly helpful for conditions like:
- Post-traumatic pain
- Post-surgical pain
- Chronic intractable pain
This technology is suitable for adults aged 18 and older, providing a new avenue for relief and a reduced reliance on traditional pain management methods.
Magstim’s Impact on Brain Disorder Research
Supporting Research in Depression and OCD
Magstim’s technology has been a go-to tool for researchers looking into conditions like depression and Obsessive-Compulsive Disorder (OCD). It’s used to explore how different parts of the brain connect and function, which is key to understanding these complex disorders. By using Magstim’s systems, scientists can investigate specific brain circuits that might be involved, helping to develop new ideas for treatments. The ability to precisely target brain areas with magnetic pulses makes it a powerful method for studying the underlying mechanisms of these conditions.
Investigating Alzheimer’s and Parkinson’s Disease
When it comes to neurodegenerative diseases such as Alzheimer’s and Parkinson’s, Magstim plays a role in research aimed at understanding how these conditions progress and how to potentially slow them down. Researchers use Magstim devices to examine brain activity and connectivity in individuals with these diseases. This helps in mapping out the changes happening in the brain over time and looking for ways to intervene. It’s all about getting a clearer picture of what’s going wrong at a neural level.
Applications in Autism and Stroke Recovery
Beyond depression and neurodegenerative conditions, Magstim’s technology is also being applied to research in autism spectrum disorder and stroke recovery. For autism, studies might look at how brain networks differ and if neuromodulation can help with certain social or communication challenges. In stroke recovery, the focus is on whether Magstim can help reawaken dormant neural pathways or support the brain’s natural ability to reorganize itself after injury. It’s a diverse area of research, showing how adaptable this technology is.
The ongoing exploration of brain disorders using Magstim’s tools highlights a shift towards understanding the brain’s intricate networks. This approach moves beyond just treating symptoms to investigating the root causes within neural circuitry. It’s a methodical process, building knowledge piece by piece.
Integration and Collaboration in Neurotechnology
Synergy with EEG and EMG Modalities
Magstim’s technology doesn’t operate in a vacuum. It’s designed to work alongside other important tools in the neurotech toolkit. Think about electroencephalography (EEG), which measures brain’s electrical activity, or electromyography (EMG), which looks at muscle electrical activity. Combining Magstim’s magnetic stimulation with these methods gives researchers a much clearer picture. You can see how the brain responds electrically to magnetic pulses in real-time. This kind of combined approach is really helpful for understanding complex brain functions and how different areas talk to each other. It’s like adding more senses to your observation.
Seamless Integration with Navigation Software
Getting the magnetic stimulation exactly where you want it in the brain is key. That’s where navigation software comes in. Magstim systems can link up with these advanced programs. This means you can plan your stimulation sessions with precision, often using imaging data like MRI scans. The software guides the coil placement, making sure you hit the right spot every time. This accuracy is a big deal for both research and making sure treatments are consistent. It takes a lot of the guesswork out of the process, which is good for everyone involved.
Fostering Collaboration Through Conferences
Innovation in neurotechnology doesn’t happen in isolation. Conferences and workshops play a big role. Magstim often participates in these events, showing off their latest gear and talking with scientists and doctors. These gatherings are great places to share ideas, discuss new research findings, and see how different technologies can work together. It’s where people connect and new projects get started. For example, events like the DGKN conference bring together folks interested in everything from TMS to high-density EEG, creating a space for shared learning and future developments in neuromodulation.
Working with other systems and sharing knowledge at events helps push the whole field forward. It means we can get a better handle on brain conditions and find new ways to help people.
The Magstim Ecosystem and Welcony Brands
Magstim isn’t just about one device; it’s part of a bigger picture under the Welcony umbrella. Think of Welcony as the parent company that brings together several specialized brands, all focused on different parts of neurotechnology. This means you get more than just magnetic stimulation. You also get access to things like MagstimEGI, which is all about high-density EEG, and other brands that handle things like nerve monitoring during surgery. It’s like a one-stop shop for brain and nerve tech.
Magstim Magnetic Stimulation
This is the core of what Magstim is known for. They’ve been doing magnetic stimulation for a long time, and their equipment is used all over the world in research and clinics. It’s the technology that helps scientists study the brain and clinicians treat conditions like chronic pain. The Magstim Rapid system, for instance, has FDA clearance for pain management, offering a non-drug, non-invasive way to help people.
MagstimEGI High-Density EEG
Beyond magnetic stimulation, Magstim also provides advanced EEG systems through MagstimEGI. These systems are designed for high-density electroencephalography, meaning they can capture very detailed brain activity. This is super useful for researchers trying to understand brain function in more detail, looking at things like sleep patterns, cognitive processes, or neurological disorders. It complements the stimulation side by allowing for detailed measurement of brain responses.
Complementary Neurotechnology Solutions
Welcony, as a whole, brings together a range of technologies that work together. Besides Magstim’s stimulation and MagstimEGI’s EEG, they also have brands like Technomed for clinical neurophysiology and Neurosign for monitoring nerves during operations. This collection of brands means that if you’re working in a hospital or a research lab, you can likely find multiple tools from Welcony that fit your needs. They aim to provide a full suite of tools for studying and treating the nervous system.
Looking Ahead
So, what does all this mean for the future? Magstim has been around for a while, over 35 years actually, and they’ve really built a name for themselves in brain stimulation tech. They’re behind a lot of the research you see out there, and their equipment is used everywhere from big research labs to regular clinics. The Magstim Rapid system, for example, is now FDA cleared for pain relief, which is a pretty big deal. It means more people can get non-drug, non-invasive help for chronic pain. They’re always updating their gear, like the new Rapid system with its fancy touchscreen and easier software. It seems like they’re really focused on making this technology more accessible and useful for both doctors and patients. It’s exciting to see where they go next with all this.
Frequently Asked Questions
What is Magstim and what does it do?
Magstim is a company that makes special machines for helping the brain and nerves. These machines use magnetic pulses to gently change how nerves work. It’s like giving your brain a little nudge to help it work better, especially for things like pain or certain brain conditions. They have been doing this for a long time and lots of scientists and doctors use their tools.
Is Magstim safe to use?
Yes, Magstim’s technology is considered safe. It’s a non-invasive method, meaning it doesn’t require surgery or needles. The U.S. Food and Drug Administration (FDA) has cleared their Magstim Rapid system for certain uses, like helping with chronic pain. Many studies have looked at how it works, and doctors and researchers trust it.
What kind of problems can Magstim help with?
Magstim machines can help with a few different things. They are used to help people with long-lasting pain that doesn’t go away easily. They are also used a lot in research to understand brain problems like depression, OCD (which is like having strong worries), Alzheimer’s, and Parkinson’s disease. Scientists are also looking into how it might help with recovery after a stroke or with autism.
How does Magstim work for pain relief?
For pain, Magstim uses magnetic pulses aimed at specific nerves. These pulses help calm down the nerves that are sending pain signals. It’s a way to reduce pain without using medicine, injections, or surgery. This can lead to less pain, better movement, and a better overall feeling for the person.
Can Magstim machines connect with other tools?
Yes, Magstim machines are designed to work well with other important tools used in studying the brain. For example, they can be used together with EEG (which measures brain waves) and EMG (which measures muscle activity). They also work with special computer programs that help guide where the magnetic pulses should go, making the treatment more precise.
What is the MagstimEGI part of Welcony?
Welcony is a company that offers different tools for brain science and health. Magstim Magnetic Stimulation is one part, focusing on magnetic pulses. MagstimEGI is another part that specializes in high-density EEG, which is a very detailed way to measure brain activity. Together, these and other Welcony brands offer a complete set of tools for studying and treating brain-related issues.